How Clean is Clean Enough? Evaluating Air Quality
Is your shop clean enough to fabricate components for medical devices? A proper diagnosis is necessary to solving an OSHA or FDA particulate problem. But before installing an air quality remedy, it helps to know your current particulate levels, exposure profile and airflow patterns. Let’s take a closer look at this evaluation process.
Posted: August 24, 2016
Is your air clean enough? Of course, the real question is: clean enough for what?
Meeting minimum OSHA regulations for indoor air quality (IAQ) is a must for every manufacturing facility. However, there are many reasons that companies may decide to go above and beyond these minimum standards. In addition to the obvious benefits for worker health, cleaner air pays dividends in worker productivity, morale, retention and recruiting. It also has another benefit: a clean facility that makes a great first impression. That can be especially important for companies fabricating components for the medical industry. If your facility isn’t making the right impression, it’s probably time to take a closer look at your air quality systems. The first step to solving a particulate problem is proper diagnosis. Before installing an air quality remedy, it helps to know your current particulate levels, exposure profile and airflow patterns. Here is a closer look at the evaluation process.
MEASURING THE PROBLEM
Visible dust on surfaces is a key indicator of air quality problems. However, to get a more accurate diagnosis of indoor air quality, you need to take some measurements. Measuring your current state will help you determine what kind of remediation is needed. Evaluation starts with measurement of dust concentrations at different points in your facility. Dust concentration meters will help to quantify your exposure risks by providing accurate measures of total particulate levels in their immediate vicinity.
If your processes generate a consistent amount of particulate at all times, it may be enough to take a few “snapshot” readings and average the result. However, if your processes are variable, you may want a unit that takes incremental measurements over a period of time to see how air quality changes over time as your industrial processes cycle. This type of analysis allows plant managers to see both “peak” measurements and total particulate exposure over the time period. Meters should be set up at breathing-zone height (roughly 5 ft off the ground) at key points around the facility, including next to the fume-generating processes and in other areas in the facility where people congregate such as workstations and aisleways. Taking measurements in a grid pattern throughout the building will help you see how fumes are propagating and where they tend to collect.
CLEANER AIR, CLEANER FACILITIES
A clean, pleasant environment is a plus for any industry, but it is especially important when the FDA knocks on your door once or twice a year. Randy B., a facilities coordinator at a medical product manufacturing company, explains: “We give frequent tours to buyers coming from hospital systems and they appreciate a clean, orderly environment. It also matters for our annual FDA audits. Ultimately, they are looking at the quality of the finished product, but if the manufacturing side looks dirty, it sends a red flag.”
As weld fumes and particulates cool, they settle out of the air and onto horizontal surfaces, leaving a thin layer of visible grime throughout the facility. This is especially problematic when particulate impacts the finishing line for medical products. Before installing their current air quality system, Randy’s plant had a problem with particulate from the welding stations contaminating their powder coat line. The particulate ruined the finish, and affected components ultimately had to be scrapped. Cleaning up the facility air fixed the problem.
CALCULATING COMPOSITION
Understanding total particulate exposure is a good first step, but composition matters, too. Exposure risks depend on the size and chemical composition of the particles generated. For example, medical parts are often made of chrome alloys, titanium or stainless steel, each of which has their own exposure profile. To ensure that you are staying under safe exposure limits for dangerous contaminants such as hexavalent chromium, manganese or beryllium, you may want to conduct additional analysis.
Air quality engineers can approximate exposure levels for specific elements by analyzing the processes and materials being used and the total particulate levels. Total exposure levels to different elements can be extrapolated from the total particulate measurements if the composition of base metals, coatings and lubricants, and consumables is well understood. If these estimates show that you are close to exposure limits on one or more elements, you may want to consider further analysis to get precise measurements. Dust samples can be collected on special filters inside the dust concentration meter. Samples are sent away to a lab for chemical analysis, a process that generally takes about two to three weeks.
UNDERSTANDING AIR FLOW
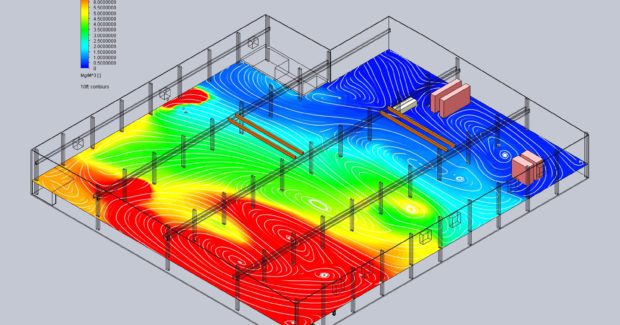
A third crucial component that air quality engineers will evaluate is how air moves through the building. Every facility has its own unique airflow patterns that influence how weld fumes propagate through the facility, where they end up, and where they linger. The dust concentration meter readings at different locations will provide important data. But designing an effective remedy also requires a more in-depth evaluation of the current airflow patterns. The type and location of existing ventilation and HVAC equipment, location of windows and doors, equipment position, temperature variations, and other factors influence airflow currents throughout the building. These currents can have a great deal of impact on the efficacy of dust capture and filtration equipment.
If you try to work against the currents, you will need a much higher capture velocity for your air filtration equipment, resulting in significantly higher energy use. Capture velocity needs are also impacted by the processes themselves. Processes that aggressively “push” particulates into the air will require much higher capture velocities than those that allow them to simply waft into the air, such as fumes rising from freshly welded parts in a cooling bin.
A CLEANER FACILITY THROUGH SCIENCE
The evaluation process allows system engineers to take a more scientific approach to air quality system design. By understanding the volume, composition and movement of particulate throughout the facility, we can make better recommendations for mitigation and select the most cost effective solution to help you reach your goals. For example, we use a computer modeling program called Ventmapping™ to analyze data gathered during evaluation, model the facility layout and airflow patterns, and try out remedy options virtually.
https://youtu.be/MFYkPNG7Cjw
So, is your facility clean enough? Sometimes you need more than your eyes to know for sure. Evaluation is the first step to creating a clean, healthy facility that will impress your employees, your customers and your OSHA or FDA inspectors.